|
4/16/2023 0 Comments Quantum dot core shell Organically passivated quantum dots have low fluorescence quantum yield due to surface related trap states. The core and the shell are typically composed of type II–VI, IV–VI, and III–V semiconductors, with configurations such as (CdS) ZnS, (CdSe) ZnS, (CdSe) CdS and (InAs) CdSe. However, the corresponding magnitudes are substantially reduced. In contrast with a dielectrically homogeneous system, dispersing the systems into a matrix with lower dielectric constant blue shifts all the peak positions and the refractive index also changes. 
Similar trend has also been observed in most of the cases when an impurity in CSQD is displaced from the core centre to the shell centre. In a CSQD it is observed that upon increasing the shell thickness there is a significant red shift in the threshold energy, also the nonlinear absorption coefficients enhance drastically and all the refractive index changes, independently on the dielectric environments. This enhancement in the quantum efficiency is due to the increased confinement of the electron and hole to the pair in the core and the dangling bonds on the surface of the QDs. This could be overcome by growing epitaxial layers of inorganic material over the QD core material to obtain a core shell quantum dot (CSQD) structure, which results in a substantial improvement in the photoluminescence efficiency of the QDs compared to their organically capped counterparts. Organic encapsulation acts as a surface trap state that aids the non-radiative de-excitation channels for the charge generated by the photon, which leads to an overall reduction in the fluorescent quantum yield. In the QD, the surface to volume ratio is high (i.e., there is a high population of atoms on the surface). Ī general strategy for obtaining and sustaining quantum confinement in a QD involves encapsulating the QD within an organic surfactant. Since this time, there has been much advancement in the synthesis of highly monodisperse quantum dots that has allowed for multitudinous spectroscopic studies of the evolution of QD as a function of their size (Fig. 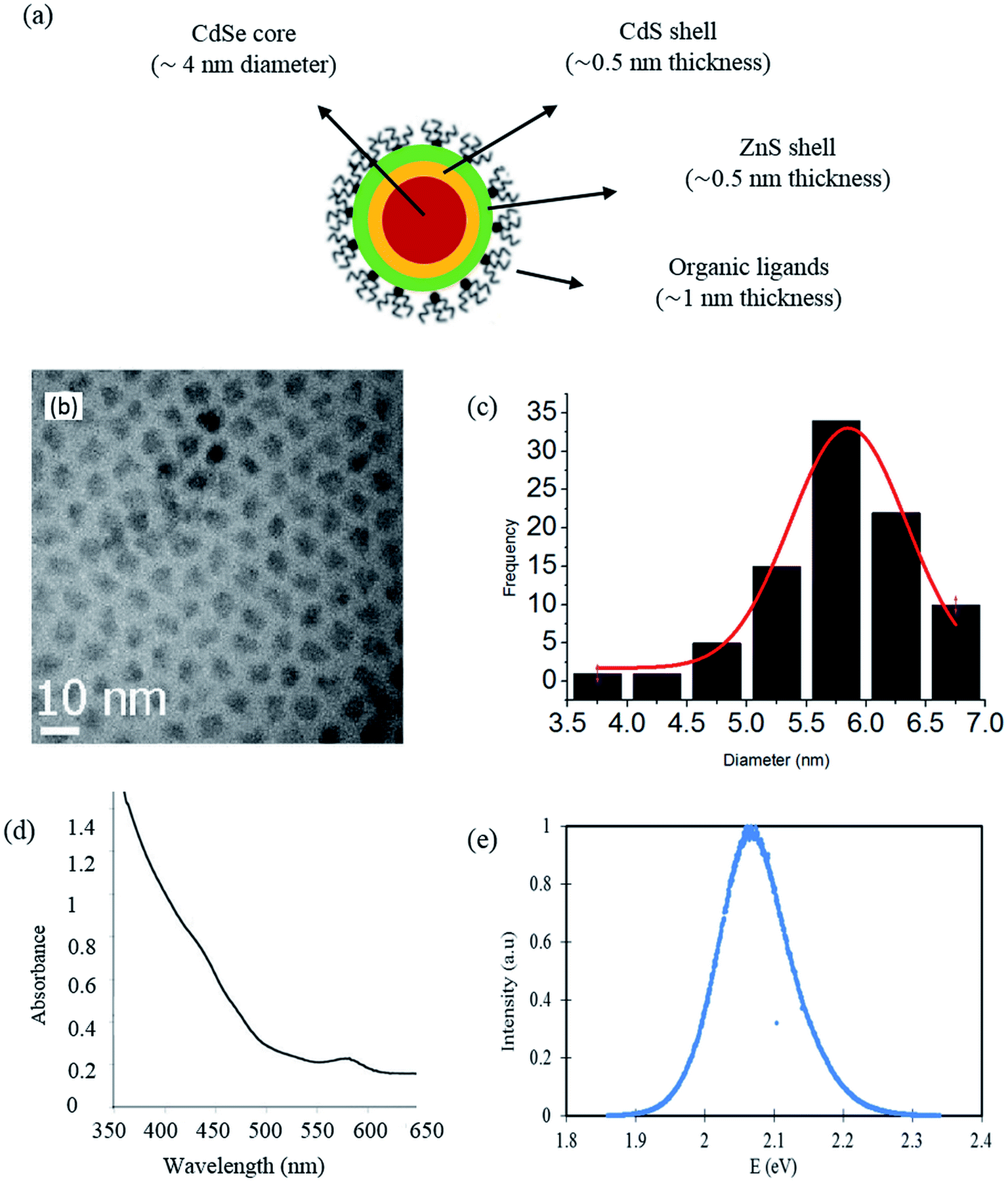
Alexei Ekimov first discovered the quantum dots in a glass matrix in the early 1980s. The electronic properties of these materials are between that of a bulk semiconductor and discrete molecules. In a QD, the electrons and holes are in a confined space in all of the three directions that results in quantisation of the electron and hole energy levels, which is considered the zero dimensional confinement effect. 
When the size decreases and becomes comparable to that of the Bohr exciton radius, the energies are discrete, which results in a large effective band gap leading to a blue shift in the optical transition compared to that of bulk materials. In the bulk, the dimensions of the semiconducting materials are much larger than the Bohr radius, and the energy levels are continuous. Excitons have a natural physical separation distance that varies from material to material, and this average separation distance is known as the Bohr exciton radius. When a weak columbic force of attraction exists between the hole and electron pair, an exciton is created. In semiconductors, when an electron leaves the valence band and enters the conduction band due to excitation, an electron–hole pair is created. They are fluorescent semiconducting nanocrystals (NCs) with a radius that is comparable to that of the Bohr exciton radius of the material. Quantum dots (QDs) are among the most researched materials that cater to the needs of present day applications, such as biolabels, , sensors, , lasers, , light emitting diodes, , and medicine. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
